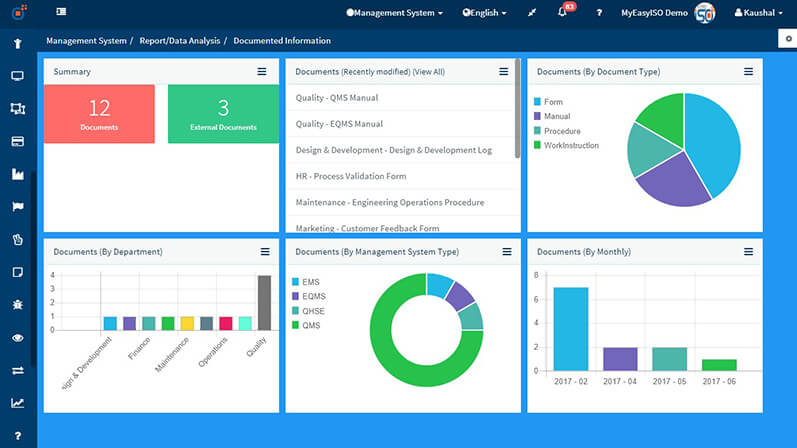
Ensuring documented information(also referred as documents and records by many users) is created, captured, documented, distributed, monitored, controlled and processed in a way that will enable all relevant interested parties and stakeholders to execute their processes quickly and correctly is very important for a successful QMS, EMS, OHSMS or any ISO management system.
A great deal of information is documented, which may include manuals, policies, SOPs, flow charts, quality plans, drawings, templates, forms, checklists, records, databases, emails, electronic data, software records, website data, software code, etc.
This documented information needs to be controlled for a robust quality management system, environment management system, occupational health & safety management system etc. wherever the standard mentions “maintain and/or retain documented information.”
The capabilities of this software ensure that you can upload a controlled document / create a new document with onscreen editor and track when it was modified or changed. Version numbers will be auto generated & incremented for each controlled change. Documents should be controlled for change and any documents of external origin need to be controlled as well as per the ISO 9001:2015 standard. Location, retention period, a disposal or archival mechanism and authority for the documented information, which needs to be retained, is also to be controlled.
The Documented Information module of Effivity acts as document control software and ensures you meet various requirements specific to documented information across the standards in an effective, efficient and simple manner. The steps below demonstrate the capabilities of this module and show how you can comply with requirements relevant to various related clauses of the standard.
Understand how Effivity can help you save more money, time and resources.
The comprehensive capabilities of this module ensure all requirements pertaining to documented information of an organisation are addressed in a quick and simplified manner while achieving 100% compliance with the documented information requirements of the ISO 9001:2015 standard, ISO 14001:2015 standard and ISO 45001:2018 standard for an effective and robust document control. Contact us at info@effivity.com for further information or visit ISOCertification/Register.htm to subscribe to our award winning and one of a kind ISO 9001, ISO 45001m ISO 14001 document control compliance software.





Trusted by Companies in over 134 Countries
Improve processes, productivity, performance & profitability with superior Quality-Health & Safety-Environment Management System