Health and Safety Management Software
Make managing health and safety easy with Effivity Health and Safety Management Software
• No card required
When it comes to health and safety, you can’t take any chances. Both the safety of your staff and the reputation of your organisation require you to plan, implement and evaluate compliant operating procedures.
Effivity’s OHSMS is automated, user-friendly, cost-effective and scalable. It removes the burden of health and safety management, allows business leaders to access relevant data at any time from anywhere and is used by auditors across the globe to ensure compliance in all aspects of large and small businesses.
Try Now
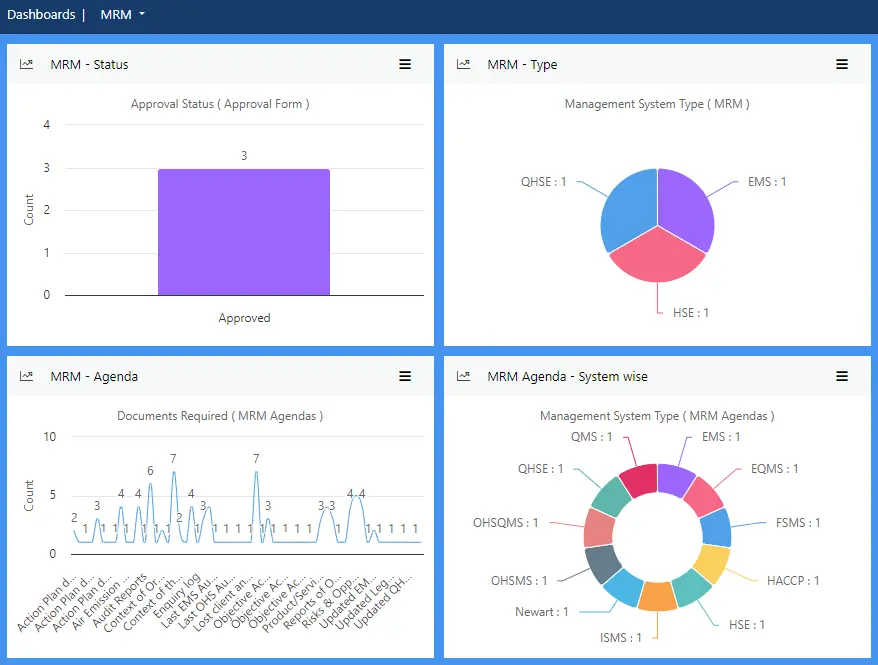
Our Occupational Health and Safety Management Software (OHSMS) enables users to adopt a proactive approach to minimising risk and reducing the number of incidents by putting all the health and safety data you need at your fingertips.
Capture, track and analyse key safety data using the simple, intuitive dashboard and enhance safety in your workplace. Effivity health and safety management software is easy to use and will help achieve and maintain ISO 45001 certification while reducing the amount of admin traditionally associated with health and safety management.
As your business grows, our health and safety software can cope with the increased management of health and safety as it’s completely scalable.
• No card required
Health and safety is key to the successful running of any business. By creating a safe working environment, the advantages of HSMS go far beyond just reducing the number of incidents.
Clear, efficient working practices make it easier to onboard new staff and train existing staff when change is necessary. Safety management software provides real-time data around staff training which helps ensure all team members have the skills and knowledge to maintain a safe workplace.
In turn, this reduces the number of incidents and amount of downtime. In addition, incidents of sickness and injury are decreased so you have a greater number of human resources at any one time.
This has a positive impact on productivity as well as helping diminish the costs associated with incident management, thus helping drive up ROI.
Paper-based safety management systems are prone to human error, illegible handwriting and misplaced documentation. All these can result in delays, misinformation, non-conformances and compliance issues.
Safety management software eliminates these issues by automating processes, providing key safety management insights and data, and making everything accessible.
Our workflow-driven software enables compliance management and corporate governance across your organisation, enabling you to become certified quickly and easily.
But it doesn’t stop there. Rules, regulations and best practices in health and safety management are constantly under review. Effivity health and safety management software ensures you remain compliant and carry out root cause analysis when issues arise. This eliminates the trial-and-error approach to mending problems and results in more efficient and cost-effective decision-making.
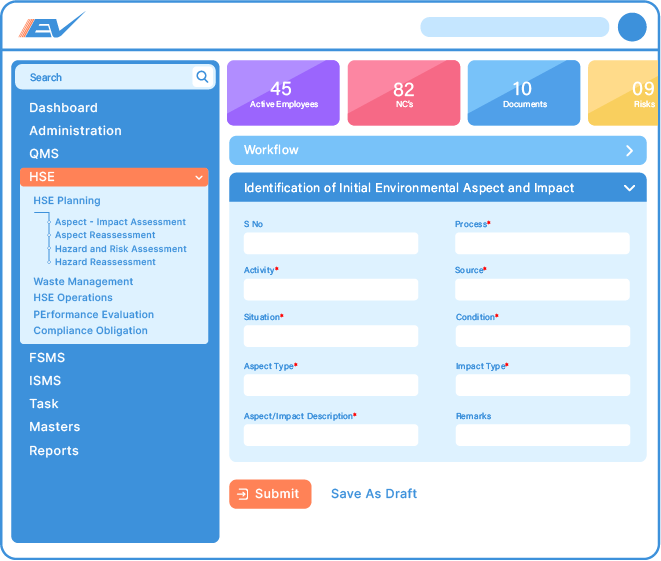
Our occupational health & safety software covers all modules required to achieve OHSMS certification as per ISO 45001:2018 standards.
✓ Higher Upfront Investments
✓ Operational Setbacks
✓ Personnel Inertia
Effivity health and safety software was designed and developed by experienced occupational health and safety specialists and delivers the same high-quality performance for businesses of all types and sizes.
It integrates seamlessly with existing systems, empowering health and safety managers. You’re in complete control and can tailor your OHSMS package to your requirements. Choose only the modules that suit your needs and pay per user, so you only pay for what you need.
As your organisation grows or adapts, you can add or remove modules to your changing circumstances using our simple-to-use drag-and-drop system. It takes just a few minutes and requires no technical knowledge or specialised training.
With Effivity, health and safety management is paperless. Everything is stored in the cloud for ease of accessibility so you can manage compliance consistently, effortlessly and in accordance with international benchmarks.
Communication is swift and easy, alerting team members to required actions, prompting follow up and preventing any tasks from slipping through the net.
If you do face any difficulties when setting up or using the Effivity OHSMS, our technical support team is on hand to help you 24/7 so you’re never alone.
Our occupational health & safety software covers all modules required to achieve OHSMS certification as per ISO 45001:2018 standards.
Carry out risk assessments for all processes and activities.Identify high-risk processes and activities and take action to minimise risks.Plan mitigation activities by preparing an operational control plan.
View more...Manage all legal, statutory, regulatory, contractual and voluntary processes and practices in one place.
View more...Successful health and safety management is the process of identifying any potential risk and taking steps to minimise the risk of harm. This is an ongoing process as new threats can arise in a fast paced world and regulations (both local and global) are apt to change.
View more...Define the boundaries of your OHSMS based on the scope for which the organisation will be certified.
View more...Good health and safety management is not just about restricting activities – it should also highlight opportunities to implement positive safety measures.
View more...Human resources are at the heart of all health and safety decisions and objectives. Creating the ideal working environment, preventing employees from coming to harm and maximising their performance are what health and safety are all about.
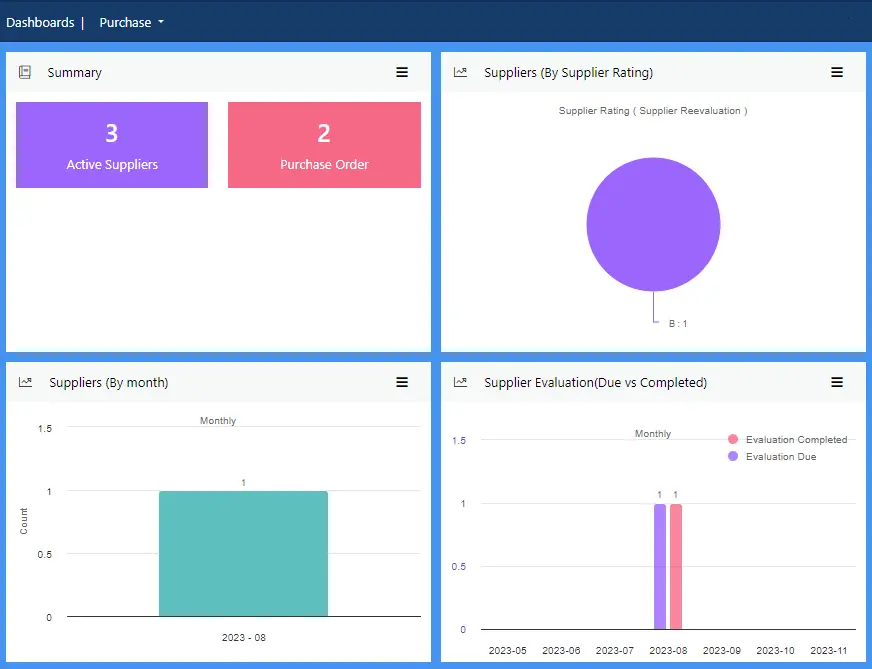
View more...Robust purchase and order practices ensure partners, raw materials and supplies give you the best chance of success.
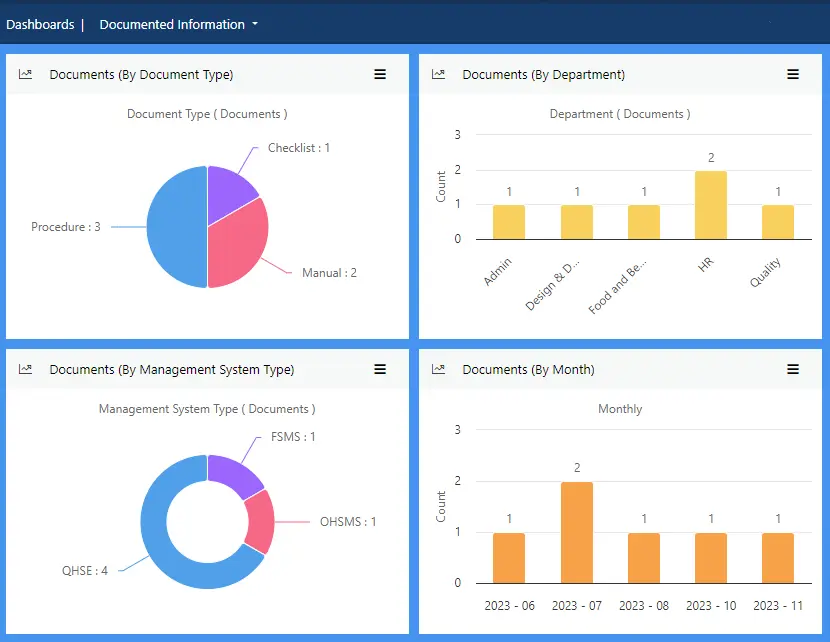
View more...Organising all your documented information ensures everything is accessible when you need it for insights into your business or for evidence of compliance.
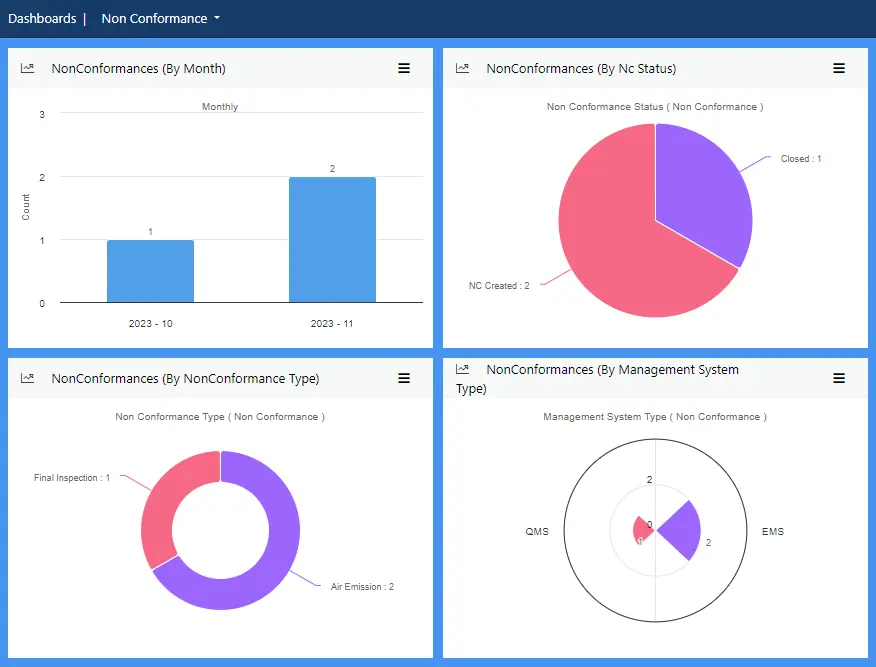
View more...Effective non-conformance management gives you a firm grip on your processes, allowing you to quickly spot issues, discover the causes and take swift remedial action.
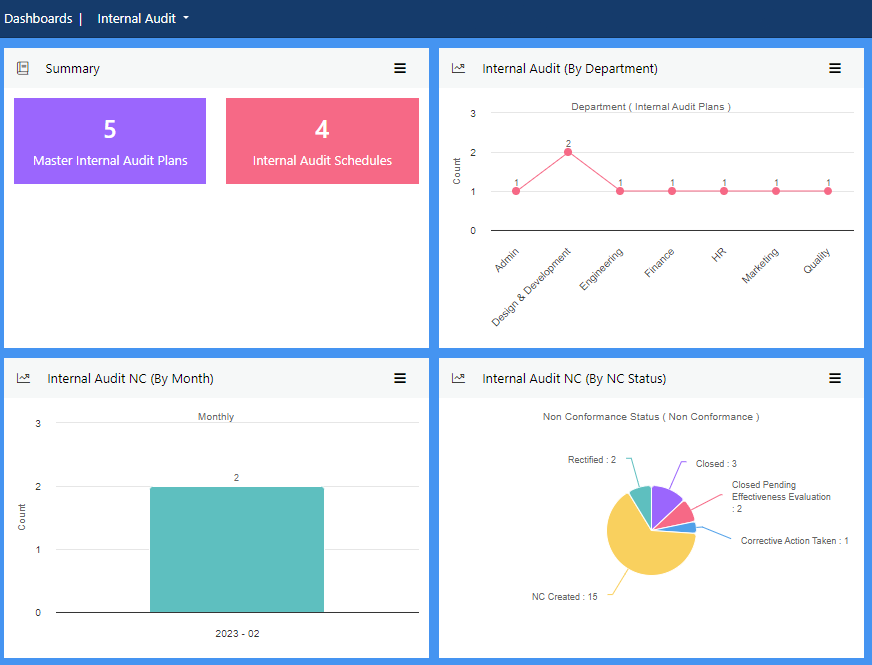
View more...The most effective way to satisfy external compliance criteria is to implement robust internal audit procedures. Good quality internal audits should focus not just on regulatory factors, but all aspects of your business. This ensures every internal audit provides a comprehensive picture of the health of each department.
View more...The Management Review Module is essential for business leaders, enabling them to quickly and easily gain actionable insights into every area of the organisation.
View more...But there are wider benefits associated with HSMS software that have an impact right across your organisation.These include: